Tag: clinical trial
Long COVID Breaks the Rules of Exercise
In the weeks after she caught COVID, in May 2022, Lauren Shoemaker couldn’t wait to return to her usual routine of skiing, backpacking, and pregaming her family’s eight-mile hikes with three-mile jogs. All went fine in the first few weeks after her infection. Then, in July, hours after finishing a hike, Shoemaker started to feel off; two days later, she couldn’t make it to the refrigerator without feeling utterly exhausted. Sure it was a fluke, she tried to hike again—and
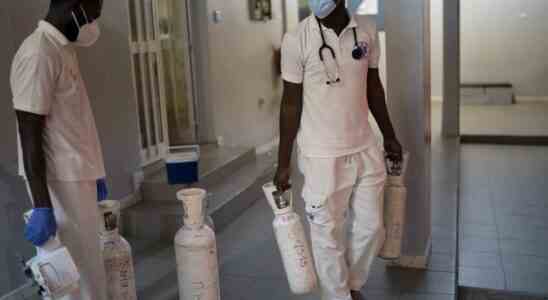
How COVID Helped Grow an Essential Treatment for Pneumonia
A version of this article was originally published in Undark Magazine.
On a late April afternoon, the Ngor Health Center in Dakar, Senegal, is serene. Sunlight spills through architectural gaps in the ceiling and lush plants line cream-colored corridors. In a patient waiting area on the second floor, a staff member gently rolls a ball back and forth with a toddler.
The calm belies the chaos at the health center eight months prior, in the summer of 2021, when COVID-19
Biden’s COVID Is Back. Is Paxlovid to Blame?
Four days after recovering from a COVID-19 infection, President Joe Biden has tested positive again. When he first got sick, Biden—like more than one-third of the Americans who have tested positive for COVID-19 this summer, according to the U.S. government’s public records—was prescribed Paxlovid, an antiviral pill treatment made by Pfizer. Like many Paxlovid takers, he soon tested negative and resumed his normal activities. And then, like many Paxlovid takers, his infection came right back. (Biden does not currently have
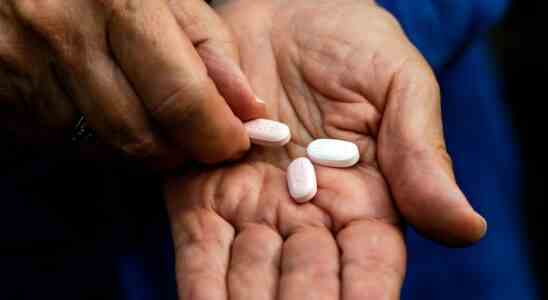
Rebound COVID Is Just the Start of Paxlovid’s Mysteries
The first data on Paxlovid, out last November, hinted that the COVID antiviral would cut the risk of hospitalization and death by 89 percent. Pundits called the drug “a monster breakthrough,” “miraculous,” and “the biggest advance in the pandemic since the vaccines.” “Today’s news is a real game-changer,” said Albert Bourla, the CEO of Pfizer, which makes the drug. The pills are “a game changer,” President Joe Biden repeated a few months later.
Now, finally, the game is being changed.
How Many Lives Could the FDA Have Saved From COVID?
The first COVID-19 vaccine could arrive before Election Day, Donald Trump avowed in the summer of 2020. But government regulators wanted things to work out differently: “The deep state, or whoever, over at the FDA is making it very difficult for drug companies to get people in order to test the vaccines and therapeutics,” he wrote on Twitter. “Obviously, they are hoping to delay the answer until after November 3rd.”
Regulators did, in fact, end up slowing the process: In